Atomic Structure Basics: Building Blocks of Life
Introduction
Understanding atomic structure basics is essential for learning chemistry and biology. Everything in the universe is made of atoms, including air, water, soil, plants, and animals. Although atoms are extremely small, they form the foundation of all matter. In fact, trillions of atoms could fit on the period at the end of a sentence. However, their tiny size does not reduce their importance. The structure of atoms determines how elements behave and how chemical reactions occur. These reactions build molecules that support life. Therefore, studying atoms helps us understand the molecular processes that sustain living organisms. You can easily download this note as a PDF using the link provided just below the post for quick access and offline reading.
Definition
Atomic structure basics describe the organization of an atom and its subatomic particles. An atom is the smallest unit of matter that maintains the properties of an element. It contains protons, neutrons, and electrons arranged in specific regions that determine the atom’s mass, charge, and chemical behavior.
Atomic Structure Basics: Components of an Atom
Atoms contain three main types of subatomic particles. Each particle has a specific role in determining the atom’s properties.
Protons
Protons are located in the nucleus, which is the central part of the atom. They carry a positive electrical charge.
- Charge: +1
- Mass: approximately 1 dalton (Da)
- Location: nucleus
The number of protons defines the identity of an element.
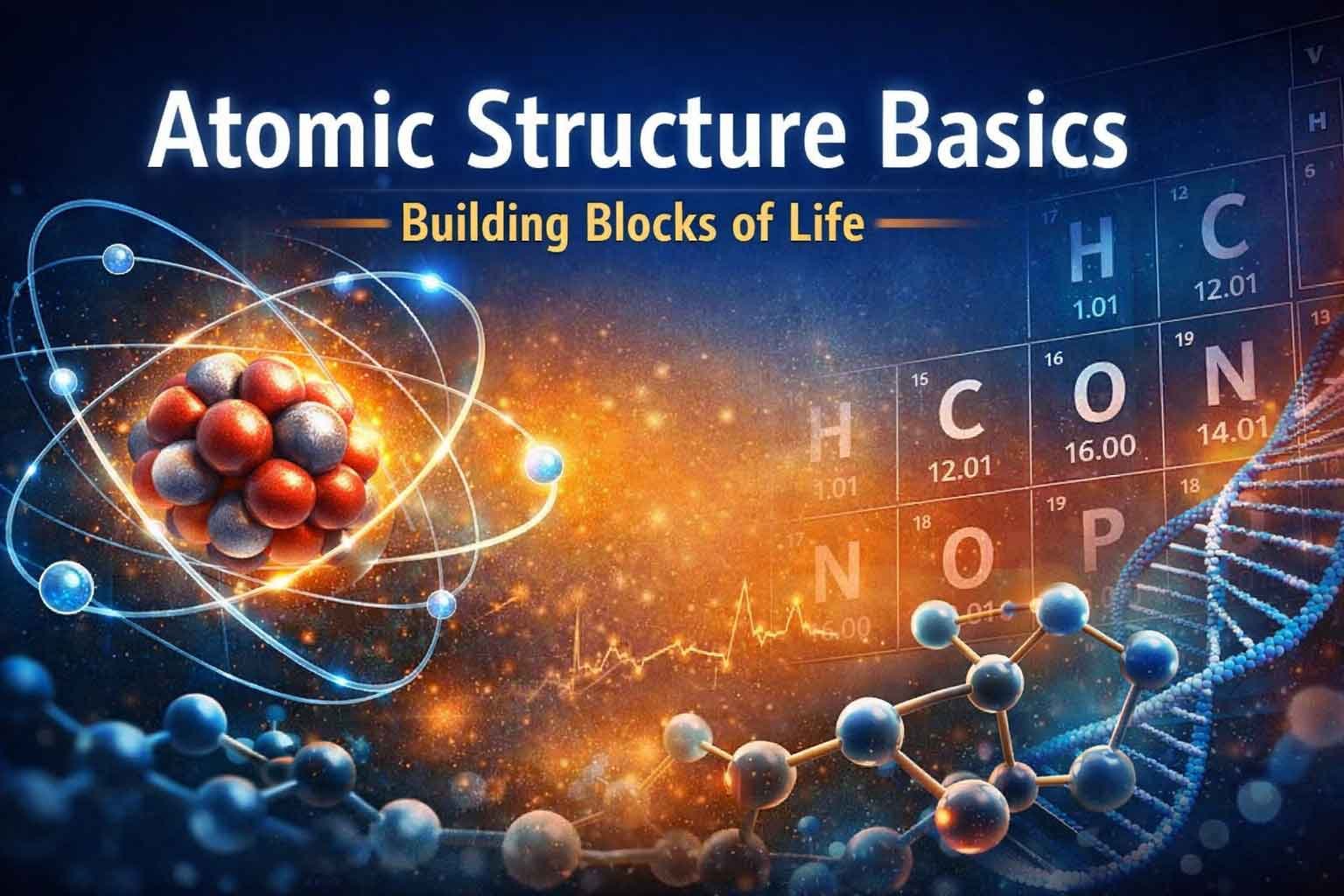
Neutrons
Neutrons also exist inside the nucleus. However, they do not carry any electrical charge.
- Charge: neutral
- Mass: approximately 1 dalton
- Location: nucleus
Neutrons contribute significantly to the mass of an atom.
Electrons
Electrons move around the nucleus in regions known as orbitals. These particles are much lighter than protons and neutrons.
- Charge: −1
- Mass: very small compared to protons
- Location: electron orbitals around the nucleus
Although electrons contribute little to atomic mass, they control chemical reactions.
Atomic Mass and the Dalton
Scientists measure the mass of atoms using a unit called the dalton (Da). This unit helps scientists compare the masses of different subatomic particles.
- Proton ≈ 1 Da
- Neutron ≈ 1 Da
- Electron ≈ 0.0005 Da
Because electrons are extremely light, they are often ignored when calculating atomic mass. As a result, the total mass of an atom mainly depends on the number of protons and neutrons.
You may also like NOTES in... BOTANY BIOCHEMISTRY MOL. BIOLOGY ZOOLOGY MICROBIOLOGY BIOSTATISTICS ECOLOGY IMMUNOLOGY BIOTECHNOLOGY GENETICS EMBRYOLOGY PHYSIOLOGY EVOLUTION BIOPHYSICS BIOINFORMATICS
Elements: One Type of Atom
An element is a pure substance composed of only one type of atom. Each element has unique chemical properties.
Common examples include:
- Hydrogen
- Oxygen
- Carbon
- Iron
The properties of an element depend on the number of protons, neutrons, and electrons present in its atoms.
Elements Essential for Life
Although many elements exist in nature, only a few are necessary for life. About 98% of living organisms are composed of six major elements:
- Carbon (C)
- Hydrogen (H)
- Oxygen (O)
- Nitrogen (N)
- Phosphorus (P)
- Sulfur (S)
In addition, several other elements play important biological roles.
- Sodium and potassium help nerve cells function properly.
- Calcium participates in cell signaling and bone formation.
- Iodine is essential for hormone production.
- Magnesium forms a part of chlorophyll in plants.
Atomic Number: Identity of an Element
The atomic number represents the number of protons present in the nucleus of an atom.
Each element has a fixed atomic number that never changes.
- Helium → 2 protons
- Carbon → 6 protons
- Oxygen → 8 protons
Because of this property, the atomic number uniquely identifies an element. Scientists arrange elements in the periodic table based on their atomic numbers and chemical properties.
Mass Number and Atomic Mass
The mass number represents the total number of protons and neutrons present in an atom.
Mass number formula:
Mass number = Protons + Neutrons
| Element | Protons | Neutrons | Mass Number |
|---|---|---|---|
| Carbon | 6 | 6 | 12 |
| Oxygen | 8 | 8 | 16 |
Because electrons are extremely light, they are usually not included in this calculation.
Isotopes: Atoms with Different Mass
Some atoms of the same element contain different numbers of neutrons. These variations are called isotopes.
Isotopes share two characteristics:
- The same number of protons
- Different numbers of neutrons
Example: Hydrogen Isotopes
| Isotope | Protons | Neutrons |
|---|---|---|
| Hydrogen | 1 | 0 |
| Deuterium | 1 | 1 |
| Tritium | 1 | 2 |
Carbon Isotopes
- Carbon-12
- Carbon-13
- Carbon-14
Most carbon atoms are carbon-12. However, smaller amounts of other isotopes also occur naturally.
Even though isotopes differ in mass, their chemical behavior is usually similar.
Atomic Weight: Average Mass of an Element
The atomic weight of an element represents the average mass of all naturally occurring isotopes of that element.
For example, hydrogen has an atomic weight of approximately 1.00794.
This value is fractional because it reflects the weighted average of different isotopes present in nature.
Radioisotopes and Radioactive Decay
Some isotopes are unstable and release energy from their nuclei. These unstable isotopes are known as radioisotopes.
During this process, atoms emit radiation such as:
- Alpha (α) particles
- Beta (β) particles
- Gamma (γ) rays
This release of energy is called radioactive decay.
Applications of Radioisotopes in Science
Tracking Organisms
Scientists sometimes feed organisms food that contains a radioisotope. Using special detectors, they can track the movement of the organism.
For example, researchers can trace the movement of earthworms in soil using this method.
Biological Tracers
Radioisotopes can be incorporated into molecules. As a result, scientists can follow these molecules inside cells or tissues.
Medical Uses
Radioactive substances also have medical applications. For instance, radiation from cobalt sources can destroy cancer cells.
However, scientists must carefully control radiation because it can damage healthy cells as well.
Electron Shells and Orbitals
Electrons move around the nucleus in regions called orbitals. These regions represent areas where electrons are most likely to be found.
Each orbital can hold a maximum of two electrons.
Orbitals are arranged into electron shells, also called energy levels.
First Electron Shell
- Contains 1 orbital
- Maximum capacity: 2 electrons
Second Electron Shell
- Contains 4 orbitals
- Maximum capacity: 8 electrons
Atoms with more electrons contain additional shells.
Electrons located farther from the nucleus have higher energy levels.
Valence Shell and Chemical Reactions
The outermost electron shell is called the valence shell.
This shell plays a crucial role in chemical reactions because it determines how atoms interact with each other.
Atoms with incomplete valence shells tend to react with other atoms. In contrast, atoms with full valence shells are chemically stable.
Examples of stable elements include:
- Helium
- Neon
- Argon
The Octet Rule
Atoms tend to form chemical bonds in order to achieve stability. In many cases, stability occurs when the outer shell contains eight electrons.
This concept is known as the octet rule.
Atoms achieve this configuration by:
- Sharing electrons
- Gaining electrons
- Losing electrons
When atoms bond together, they form molecules. These molecules create the complex chemical structures necessary for life.
Conclusion
Understanding atomic structure basics helps explain how matter behaves at the molecular level. Atoms contain protons, neutrons, and electrons arranged in a structured system. These particles determine atomic identity, mass, and chemical behavior.
Furthermore, concepts such as isotopes, electron shells, and valence electrons explain how atoms interact with each other. By learning atomic structure basics, students can better understand chemical bonding, molecular formation, and the biological processes that sustain life.
Frequently Asked Questions (FAQs)
1. What are atomic structure basics?
Atomic structure basics refer to the arrangement of subatomic particles inside an atom. An atom contains protons, neutrons, and electrons. These particles determine the atom’s mass, charge, and chemical properties.
2. What are the three main parts of an atom?
The three main subatomic particles in an atom are:
- Protons (positively charged)
- Neutrons (neutral)
- Electrons (negatively charged)
Protons and neutrons are found in the nucleus, while electrons move in orbitals around the nucleus.
3. Why are electrons important in chemical reactions?
Electrons determine how atoms interact with each other. Chemical reactions occur when electrons are transferred or shared between atoms. Therefore, electrons control the formation of chemical bonds and molecules.
4. What is the difference between atomic number and mass number?
The atomic number represents the number of protons in an atom and identifies the element. The mass number represents the total number of protons and neutrons present in the atom.
5. What are isotopes?
Isotopes are atoms of the same element that contain the same number of protons but different numbers of neutrons. Because of this difference, isotopes have different masses but usually show similar chemical behavior.
6. What is the valence shell?
The valence shell is the outermost electron shell of an atom. Electrons present in this shell determine how an atom reacts with other atoms and forms chemical bonds.
7. Why is atomic structure important in biology?
Atomic structure is important because atoms combine to form molecules such as proteins, carbohydrates, and DNA. These molecules support essential biological processes in living organisms.
<<< Back to Chemistry for Biologists Page
You may also like... NOTES QUESTION BANK COMPETITIVE EXAMS. PPTs UNIVERSITY EXAMS DIFFERENCE BETWEEN.. MCQs PLUS ONE BIOLOGY NEWS & JOBS MOCK TESTS PLUS TWO BIOLOGY PRACTICAL
Study Offline!! Download this Note as a PDF
🌿 Dear Readers,
I hope you found this article helpful and easy to understand. If you have any questions, suggestions, or thoughts, I would truly love to hear from you.
Please share your feedback in the comments below. Your participation helps make EasyBiologyClass a better learning space for everyone.
Best regards,
EasyBiologyClass